Top UN Officials Resign Over The Organization’s Incapability To Protect Human Rights In Palestine
November 30, 2023
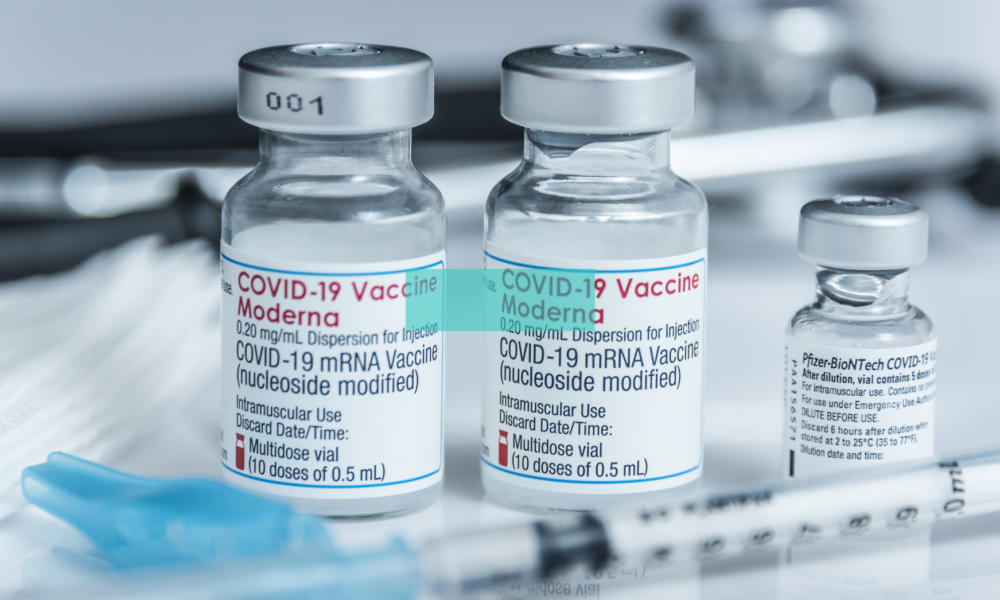
July 11 (Reuters) – Moderna Inc said on Monday it was advancing two Omicron vaccine candidates for the fall, one designed against the BA.1 variant and another against the BA.4 and BA.5.
Vaccine makers including Moderna and rival Pfizer Inc are developing updated vaccines to target the fast-spreading Omicron subvariants BA.4 and BA.5, which have gained a foothold in the United States over recent weeks.
Moderna said its decision to develop the bivalent vaccines was based on different market preferences for shots against the subvariants.
Bivalent vaccines are designed to target two different coronavirus variants – the original variant from 2020 and the newer Omicron variants.
Moderna said new clinical data for its mRNA-1273.214 vaccine, designed to target the BA.1 variant, showed significantly higher neutralizing antibody responses against the fast-spreading BA.4 and BA.5 compared with the currently authorized booster.
The company’s second booster candidate, mRNA 1273.222, is based on the BA.4/5 strain and is being developed in accordance with recent U.S. Food and Drug Administration advice.
Jefferies analyst Michael Yee said though the new data on the company’s BA.1 subvariant-targeting booster shows better durability and antibody response, the original Omicron variant has already shifted in the United States and the FDA is likely to approve planned BA.4/5 boosters. (Reporting by Mrinalika Roy and Leroy Leo in Bengaluru; Editing by Devika Syamnath)